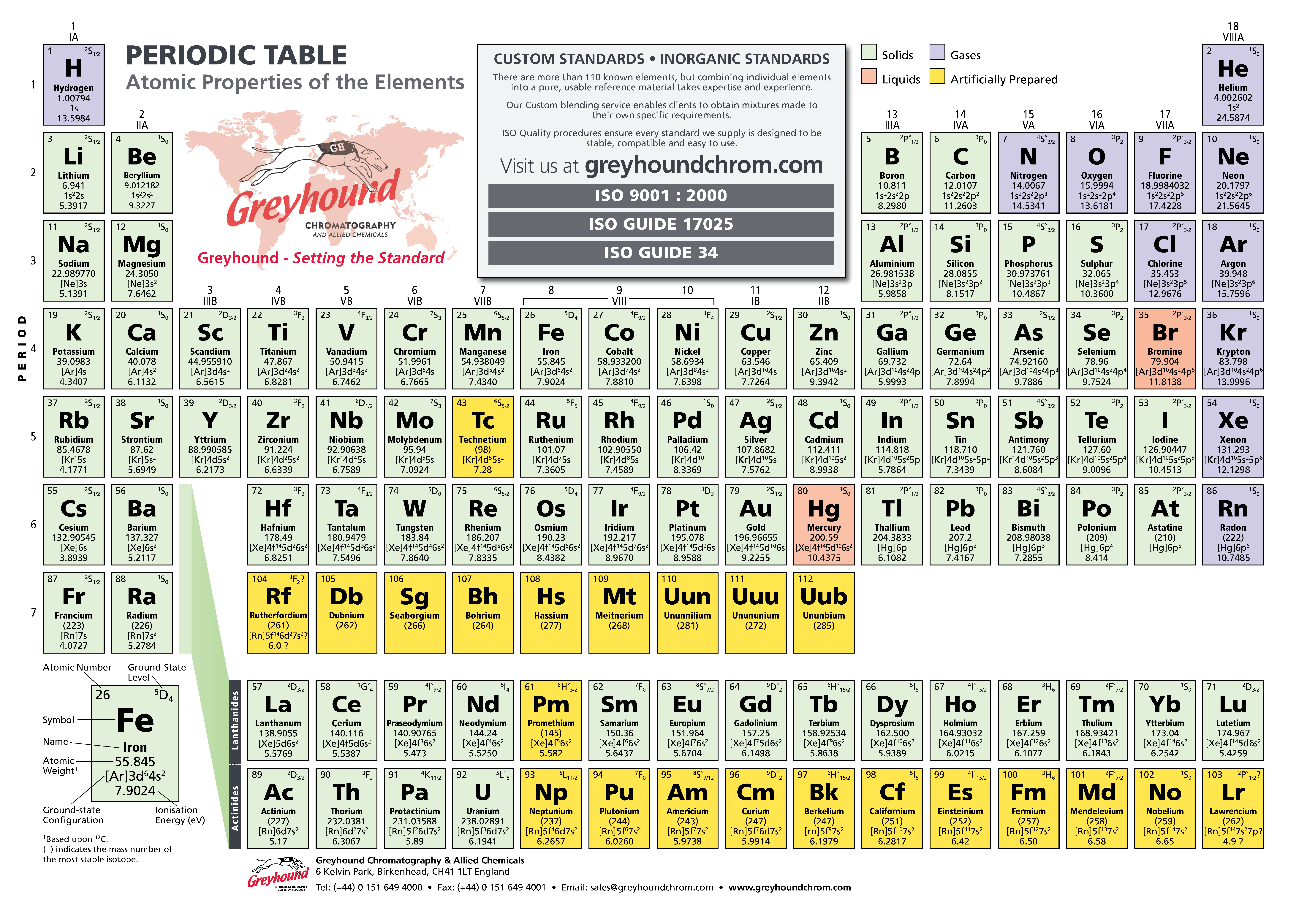
Greyhound Chromatography Periodic Table
The periodic table of the chemical elements (also periodic table of the elements) is a tabular display of the chemical elements. Although precursors to this table exist, its invention is generally credited to Russian chemist Dmitri Mendeleev in 1869, who intended the table to illustrate recurring periodic trends in the properties of the elements. The layout of the table has been refined and extended over time, as new elements have been discovered, and new theoretical models have been developed to explain chemical behaviour.
Over 70 High Purity Standards inc. single element standards are available from Greyhound Chromatography and Allied Chemicals. Concentrations include both 1000 and 10.00 in aqueous solution, (unless otherwise noted). Most standards are packaged in 50,100, 250 and 500 mL HDPE or LDPE laboratory grade bottles. The density is provided on the Certificate of Analysis as additional information for the user.
The accuracy of all standards is verified against NIST Spectrometric Standard Solutions. A certificate of Analysis and Safety Data Sheet are included with each standard. Standards are certified accurate for a period of 18 months from the date of shipment unless stated otherwise on the Certificate of Analysis.
The periodic table is now ubiquitous within the academic discipline of chemistry, providing a useful framework to classify, systematise, and compare all of the many different forms of chemical behavior. The table has found many applications in chemistry, physics, biology, and engineering, especially chemical engineering.
In 1789, Antoine Lavoisier published a list of 33 chemical elements. Although Lavoisier grouped the elements into gases, metals, non-metals, and earths, chemists spent the following century searching for a more precise classification scheme. In 1829, Johann Wolfgang Döbereiner observed that many of the elements could be grouped into triads (groups of three) based on their chemical properties. Lithium, sodium, and potassium, for example, were grouped together as being soft, reactive metals. Döbereiner also observed that, when arranged by atomic weight, the second member of each triad was roughly the average of the first and the third. This became known as the Law of triads. German chemist Leopold Gmelin worked with this system, and by 1843 he had identified ten triads, three groups of four, and one group of five. Jean Baptiste Dumas published work in 1857 describing relationships between various groups of metals. Although various chemists were able to identify relationships between small groups of elements, they had yet to build one scheme that encompassed them all.
German chemist August Kekulé had observed in 1858 that carbon has a tendency to bond with other elements in a ratio of one to four. Methane, for example, has one carbon atom and four hydrogen atoms. This concept eventually became known as valency. In 1864, fellow German chemist Julius Lothar Meyer published a table of the 49 known elements arranged by valency. The table revealed that elements with similar properties often shared the same valency.
Of the 94 naturally occurring elements, those with atomic numbers 1 through 40 are all considered to be stable isotopes. Elements with atomic numbers 41 through 82 are apparently stable (except technetium and promethium) but theoretically unstable, or radioactive. The half lives of elements 41 through 82 are so long however that their radioactive decay has yet to be detected by experiment. These theoretical radionuclides have half lives at least 100 million times longer than the estimated age of the universe. Elements with atomic numbers 83 through 94 are unstable to the point that their radioactive decay can be detected. Some of these elements, notably thorium (atomic number 90) and uranium (atomic number 92), have one or more isotopes with half lives long enough to survive as remnants of the explosive stellar nucleosynthesis that produced the heavy elements before the formation of our solar system. For example, at over 1.9×1019 years, over a billion times longer than the current estimated age of the universe, bismuth-209 (atomic number 83) has the longest known alpha decay half life of any naturally occurring element. The very heaviest elements (those beyond plutonium, atomic number 94) undergo radioactive decay with half lives so short that they have only been observed as the result of experimental observation.
Apart from the hydrogen and helium in the universe, which are thought to have been mostly produced in the Big Bang, the chemical elements are thought to have been produced by one of two later processes—either cosmic ray spallation (important for lithium, beryllium and boron), or stellar nucleosynthesis (which produces all elements heavier than boron). Oxygen is the most abundant element in the Earth's crust, making up almost half of its mass Relatively small amounts of elements continue to be produced on Earth as products of natural transmutation processes. This includes production by cosmic rays or other nuclear reactions (see cosmogenic and nucleogenic nuclides), or as decay products of long-lived primordial nuclides.
English chemist John Newlands produced a series of papers in 1864 and 1865 that described his own classification of the elements: he noted that when listed in order of increasing atomic weight, similar physical and chemical properties recurred at intervals of eight, which he likened to the octaves of music. This law of octaves, however, was ridiculed by his contemporaries and the Chemical Society refused to publish his work. Nonetheless, Newlands was able to draft an atomic table and use it to predict the existence of missing elements, such as germanium.
Russian chemistry professor Dmitri Ivanovich Mendeleev and Julius Lothar Meyer independently published their periodic tables in 1869 and 1870, respectively. They both constructed their tables in a similar manner: by listing the elements in a row or column in order of atomic weight and starting a new row or column when the characteristics of the elements began to repeat. The success of Mendeleev's table came from two decisions he made: The first was to leave gaps in the table when it seemed that the corresponding element had not yet been discovered. Mendeleev was not the first chemist to do so, but he was the first to be recognised as using the trends in his periodic table to predict the properties of those missing elements, such as gallium and germanium.
The second decision was to occasionally ignore the order suggested by the atomic weights and switch adjacent elements, such as cobalt and nickel, to better classify them into chemical families. With the development of theories of atomic structure, it became apparent that Mendeleev had listed the elements in order of increasing atomic number.
CONTACT US
Tel: +44 (0) 151 649 4000
Email: marketing@greyhoundchrom.com
FOLLOW US
YOU MAY ALSO BE INTERESTED IN OUR NEWSLETTER